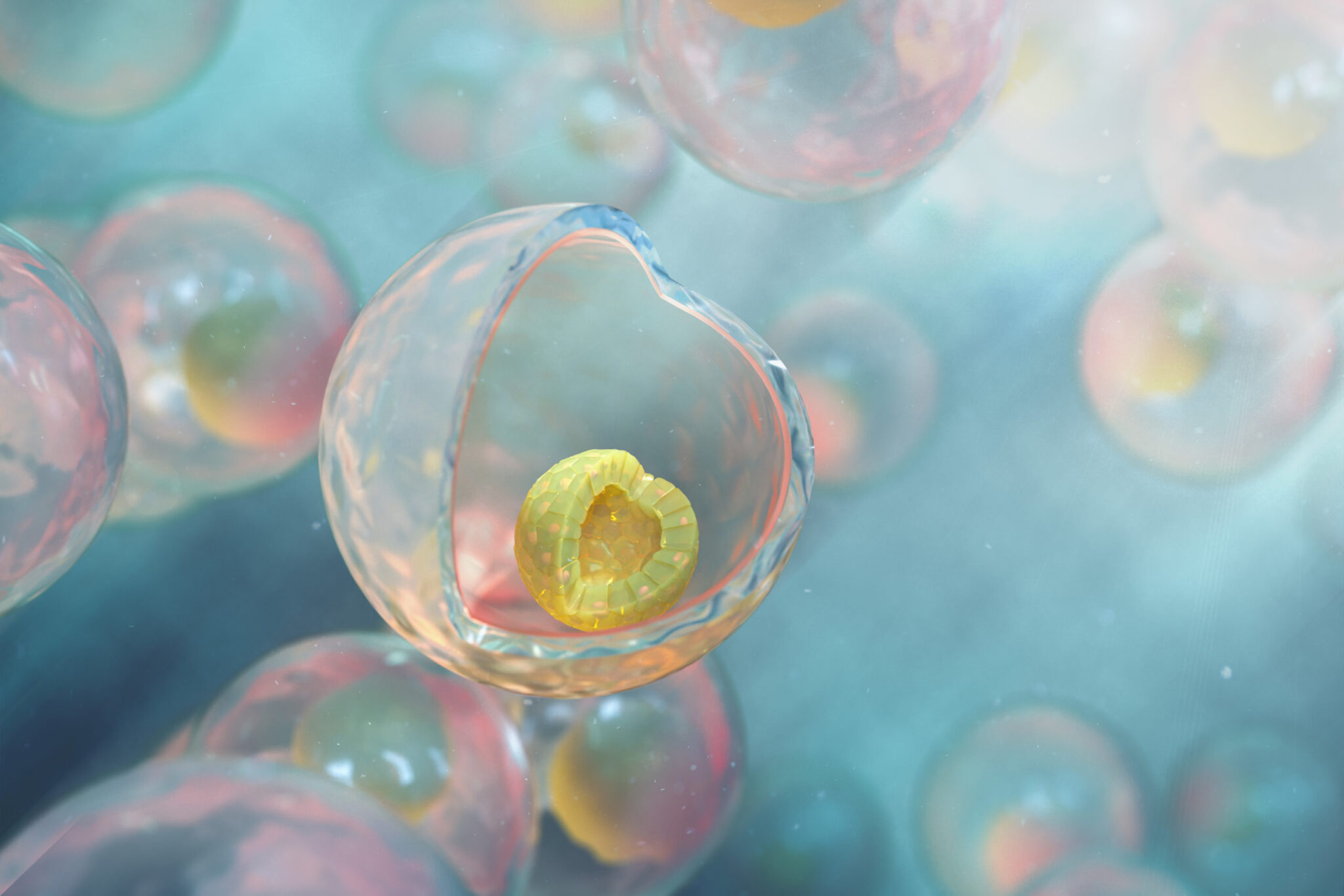
TreeFrog Therapeutics will apply its proprietary technology for the mass-production of stem cell therapies to the world-first single-step protocol allowing the differentiation of human induced pluripotent stem cells (iPSCs) into fully functional human hematopoietic stem cells (HSCs). Resulting off-the-shelf universal HSC grafts could reduce costs and waiting times for patients in need of a bone marrow transplant.
Bordeaux, France – November, 17th, 2020. The French Blood Agency (EFS) and TreeFrog Therapeutics announce a partnership to advance the mass-production of iPSC-derived HSC transplants based on a patented differentiation protocol developed by the EFS (UMR S938), the Institute for Radioprotection and Nuclear Safety (IRSN) and Sorbonne University[1]. Under the terms of the agreement, TreeFrog Therapeutics will demonstrate its proprietary C-StemTM technology enables the production of large batches of iPSC-derived HSCs in industrial bioreactors with clinical-grade quality and reduced costs compared to 2D culture.
Hematopoietic stem cell transplantation: over $100,000 per patient
In 2018, over 24,000 HSC transplants were performed in the US (CIBMTR data) and over 45,000 in Europe (EMBT data) to restore hematopoiesis and immune function in patients suffering from leukemias, lymphomas, anemias, hemoglobinopathies and auto-immune diseases. The global market for HSC transplantation was valued at $3,7 billion in 2015, with a CAGR of 10,6% (Credence Research data). Autologous HSC transplantations – using the patient’s own HSCs – now represent 60% of procedures, with total medical costs of $140,000 per patient. Allogeneic transplantation is performed in 40% of cases. This risky procedure, even more expensive (over $250,000 per patient), requires the availability of HSCs from a compatible donor and the administration of immunosuppressive medication to prevent graft-versus-host disease.
The world-first single-step differentiation protocol to differentiate iPSCs into HSCs
In 2017, French researchers from the EFS, IRSN and Sorbonne University patented the first differentiation protocol allowing for the generation of fully functional HSC transplants from iPSCs in a single step.
“Over the past 6 years, we have tested our protocol with 5 iPSC lines and demonstrated full hematopoietic recovery in large-scale in vivo studies (N=400). Our iPSC-derived HSC transplants are now well-characterized. All we need to advance our product is a scalable manufacturing process to produce large-size batches with reduced costs”
Dr Alain Chapel, Principal Investigator, IRSN.
C-StemTM: a 3D technology to mass-produce iPSC-based cell therapies
With the view of producing clinical-size batches of HSC transplants, the EFS turned to TreeFrog Therapeutics. The French startup has developed C-StemTM, a proprietary technology combining high-throughput stem cell encapsulation (1,000 capsules per second) with the scale-up potential of industrial bioreactors.
“The obvious benefit of the alginate capsule is to protect cells from shear-stress in bioreactors, which allows for the amplification and differentiation of large batches of iPSCs with reduced labor costs. But I believe the main benefit of C-Stem lies in the 3D epiblastoid conformation adopted by stem cells inside the capsule. By recapitulating the in vivo stem cell niche, it promotes high-fidelity chromosome segregation and maintains homogenous pluripotency, therefore yielding high-quality, ready-to-graft functional microtissues upon differentiation. Partnering with a patient-centered organization such as the EFS with a long-standing experience in HSC transplants is a tremendous opportunity to bring the benefits of C-StemTM to the clinic, in an indication where safety and affordability are critical”.
Maxime Feyeux, President and Chief Scientific Officer, TreeFrog Therapeutics.
“Our research teams have been working since 2014 with the aim of creating ready-to-use universal iPSC-derived HSC transplants. Through the partnership with TreeFrog, our goal is to scale-up this therapeutic approach, standardizing quality and reducing costs, so that we can provide patients waiting for an HSC transplantation with a readily available solution, with reduced risks of graft-versus-host disease.”
Dr Laurence Guyonneau-Harmand, EFS Scientist
[1] Patent EP 187113667 EFS / INSERM / SORBONNE UNIVERSITÉ / UPSud / CNRS / IRSN
About the French Blood Agency (EFS)
Thanks to blood donations, donors and recipients are at the heart of the sharing economy. The 10,000 employees, doctors, pharmacists, biologists, laboratory technicians, researchers, nurses and collection teams of the French Blood Agency, together with thousands of volunteers, are committed to transforming donations into lives and treating one million patients a year, both on a daily basis and in exceptional circumstances. The EFS is the only operator of blood transfusion in France, and its core mission is national self-sufficiency in blood products. It is also involved in many other activities, such as medical biology examinations, cell and tissue therapies and research. Beyond the testing of blood donations, EFS’ laboratories carry out different types of biological, haematological and immunological tests. These tests are essential in the context of a transfusion but also for organ, tissue or cell transplants. With more than 500 million examinations performed each year, the EFS is the largest medical biology laboratory in France. The French Blood Establishment is present all over the country to be as close as possible to donors and patients.
French Blood Agency. Much more than blood donation.
About TreeFrog Therapeutics
TreeFrog Therapeutics is a startup company aiming at providing access to cell therapies for millions of patients. TreeFrog Therapeutics has developed C-StemTM, a high-throughput cell encapsulation technology allowing for the mass-production and differentiation of stem cells in industrial bioreactors. This proprietary technology platform provides an end-to-end and scalable solution that dramatically improves the quality of therapeutic cells and reduce treatment costs.
Since its incorporation in November 2018 in Bordeaux, France, TreeFrog Therapeutics has experienced strong acceleration, raising over €13M ($14.65M) in 2019. In January 2020, TreeFrog Therapeutics joined the FrenchTech120, an elite programme for the fastest-growing technology companies in France. The team – currently 30+ employees – moved into a brand-new production facility of 13,000 sq ft in June 2020. In October 2020, TreeFrog Therapeutics was awarded the Prix Galien MedStartup for its international consortium on iPSC quality with the Harvard Stem Cell Institute and the Dana Farber Cancer Institute in Boston (USA), the FRBI in Kobe (Japan) and the Imagine Institute in Paris (France).

TreeFrog Therapeutics is receiving funding from the European Union’s Horizon 2020 research and innovation program through the SME Instrument Phase 2 under grant agreement n° SME 881113.
Press Contacts
EFS
Lola Terrasson – lola.terrasson@efs.sante.fr – +336 7210 7106
TreeFrog Therapeutics
Pierre-Emmanuel Gaultier – pierre@treefrog.fr – +336 4577 4258
Download 3D picture
Caption: Representation of human induced pluripotent stem cells self-organized in 3D within an alginate capsule produced with C-StemTM – TreeFrog Therapeutics’ proprietary technology to scale-up hiPSC-derived cell therapy manufacturing.
Credits : TreeFrog Therapeutics