La technologie biomimétique de TreeFrog Therapeutics repousse les limites de la production industrielle de thérapies cellulaires issues de cellules souches. La société vient de produire un lot de 15 milliards de cellules souches, avec une croissance exponentielle inédite dans des bioréacteurs de 10 litres, tout en maintenant un haut niveau de qualité.
Bordeaux, France – 13 avril 2021. La biotech française de thérapie cellulaire TreeFrog Therapeutics annonce être parvenue à cultiver des cellules souches pluripotentes induites humaines (hiPSC) dans un bioréacteur de 10 litres, avec un facteur d’amplification sans précédent. Grâce à sa technologie propriétaire C-StemTM , TreeFrog Therapeutics a produit un lot de 15 milliards de cellules en 6,5 jours en partant de 50 millions de cellules. La société rapporte également une qualité cellulaire exceptionnelle. Les données seront présentées lors de l’International Society for Stem Cell Research (ISSCR) Virtual Meeting, qui se tiendra du 21 au 26 juin 2021.
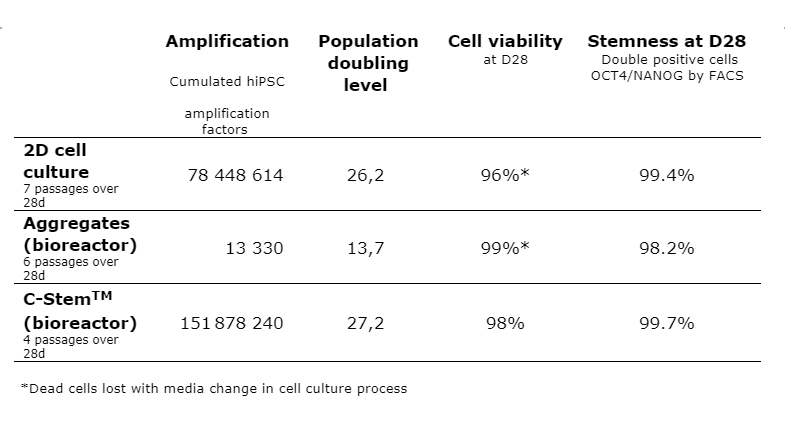
A scalable approach to overcome the limitations of 2D cell culture
With a 151 millionfold cumulated amplification, C-Stem™ nearly doubles the performance of 2D culture.
“Traditionally, pluripotent stem cells are grown in 2D, at the bottom of Petri dishes,” said Maxime Feyeux, co-founder and Chief Scientific Officer of TreeFrog Therapeutics. “And while 2D enabled the launch of the first trials with iPS-derived therapeutic cells, this technology now constitutes a critical bottleneck for cell therapy manufacturing because of its inherent limitations in scaling. 2D cell culture is expensive and lacks reproducibility at a commercial scale because of the labor, time, and space it requires. For instance, to produce the batch of 15 billion cells that we recently announced, it would require ramping to 4 full-time staff over 2 weeks and hundreds of Petri dishes, whereas it took us 2 staff for less than a week and a single 10L bioreactor with C-Stem™.”
Scale-up in bioreactors – C-Stem™ leapfrogs aggregate culture
Over 28 days, a 13,330-fold amplification was observed with hiPSC aggregates in bioreactors, with six passages, while a 151 millionfold amplification was obtained with C-Stem™ in four passages.
“I believe two core factors explain such a difference in yield,” said Kevin Alessandri, co-founder and Chief Technical Officer of TreeFrog Therapeutics. “On the one hand, our cell encapsulation technology is protecting the cells from impeller-induced shear stress in bioreactors, therefore limiting in-process cell death. On the other, the biomimetic microenvironment that we engineered inside the capsule allows hiPSCs to self-organize into an empty-core epiblast-like structure promoting fast and high-fidelity cell division, thus contrasting with the necrotic core found in aggregates.”
Benefits in genomic quality to be further investigated
The benchmark data shows that final cell viability and pluripotency are comparable across the different cell culture systems.
“However, our preliminary results suggest that C-Stem™ is the only technology with the potential of maintaining genomic integrity throughout large-scale hiPSC expansion,” said Feyeux. “In the coming months, we’ll investigate the benefits of C-Stem™ with the QC-Stem Consortium, which brings together experts in clinical- grade hiPSC quality control from the Harvard Stem Cell Institute in Boston, USA, the FBRI in Kobe Japan, and the Imagine Institute in Paris, France.”
About TreeFrog Therapeutics
TreeFrog Therapeutics is a startup company aiming at providing access to cell therapies for millions of patients. TreeFrog Therapeutics has developed
C-Stem™, a high-throughput cell encapsulation technology allowing for the mass-production and differentiation of stem cells in industrial bioreactors. This proprietary technology platform provides an end-to-end and scalable solution that dramatically improves the quality of therapeutic cells and reduce treatment costs.
Since its incorporation in November 2018 in Bordeaux, France, TreeFrog Therapeutics has experienced strong acceleration, raising over €13M ($15.60M) in 2019. In January 2020, TreeFrog Therapeutics joined the FrenchTech120, an elite programme for the fastest-growing technology companies in France. The team—currently 40+ employees—moved into a brand-new production facility of 13,000 sq ft in June 2020. In October 2020, TreeFrog Therapeutics was awarded the Prix Galien MedStartup for its international consortium on iPSC quality with the Harvard Stem Cell Institute, the FRBI in Kobe (Japan) and the Imagine Institute in Paris (France).

TreeFrog Therapeutics is receiving funding from the European Union’s Horizon 2020 research and innovation program through the SME Instrument Phase 2 under grant agreement n° SME 881113.
Press Contacts
TreeFrog Therapeutics
Pierre-Emmanuel Gaultier – pierre@treefrog.fr – +336 4577 4258
